The central nervous system is the most complicatedly and precisely regulated organ in vertebrate. It is also critical for both primary nervous activity, such as sense perception, emotion regulation and body maintenance, and higher nervous activity, such as thinking, cognition and consciousness. One of the most important traits of brain is the storage of massive information, i.e. the ability and process of learning and memory, and its malfunction, however, is the most significant clinical feature among the patients with human mental disorders, like Alzheimer's disease.
Neural synapses, the connection units between neural cells, and the information transmission among them, are the most basic and unique character of the neural system. They are also the bases for the neural circuit performing physiological functions, and their deregulation is one of the most leading causes for the neurological diseases.
The research progresses in neuroscience field have indicated that the change of the strength of synaptic transmission is the material basis for learning and memory, and these studies lead to propose the theory of “synaptic plasticity”, in which the synaptic connections strength between neurons are modulated according to the change of their neuronal activities.
Long-term potentiation (LTP) is a typical and critical form of synaptic plasticity and is also regarded as an important cellular and molecular model to study learning and memory. Currently, most of the studies in LTP field are focused on the trafficking mechanisms of extrasynaptic excitatory glutamate receptor AMPAR (α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor), while the underlying postsynaptic molecular basis is still unclear. To study this question, the “Neural Synaptic Mechanism and Function” group from Kunming Institute of Zoology, Chinese Academy of Sciences leaded by Dr. SHENG Nengyin, collaborating with Dr. Roger Nicoll Lab from University of California, San Francisco, had set up the conditional knock-out mice of all the three genes encoding AMPARs as the model system.
The researchers constructed a tethered AMPAR of GluA1 receptor with its auxiliary subunit TARP (transmembrane AMPA receptor regulatory protein) g-8, named GluA1-g-8, and then replaced the endogenous AMPARs through in utero electroporation, thereby to study the function and mechanism of the interaction of glutamate receptor complex and PDZ domain-contained postsynaptic scaffolding proteins during LTP. PDZ domain is a common structural domain of 80-90 amino-acids found in the signaling proteins of bacteria, yeast, plants, viruses and animals.
Combining the techniques of hippocampal slice culture and electrophysiology, as well as many others, they found that the trafficking of exogenous expressed AMPARs is only regulated by the tethered g-8 protein. Moreover, the PDZ-binding site-mediated interaction, between AMPAR/TARP receptor complex and postsynaptic scaffolding proteins, is necessary for the synaptic basal transmission and LTP expression of this receptor complex. Further study also demonstrated that the interaction with the PDZ-domain of postsynaptic scaffolding proteins is also required for LTP of the receptor complex of KAR (kainite receptor) with its auxiliary subunits Neto proteins, another subfamily member of glutamate receptor.
The results of this study suggested that the postysynaptic mechanism underlying LTP is conserved and regulated by a general mechanism, regardless of the subtype of glutamate receptors. Moreover, a working model is proposed that the PDZ domain-contained scaffolding proteins are the targets for modification during LTP, whereas the glutamate receptor/auxiliary subunit complexes play passive roles.
This study has revealed the postsynaptic molecular mechanism underlying synaptic plasticity LTP, which provide important theoretical basis to further study learning and memory, as well as the pathological mechanism of related mental disorders. The results have been published online on the journal of Proceedings of the National Academy of Sciences (PNAS) with the title of “LTP requires postsynaptic PDZ-domain interactions with glutamate receptor/auxiliary protein complexes”, on March 26, 2018. Dr. SHENG Nengyin is the first author and the co-corresponding author, together with Dr. Roger Nicoll. This work was funded by grants from the Strategic Priority Research Program of the Chinese Academy of Sciences, National Natural Science Foundation of China and the Chinese Academy of Sciences Pioneer Hundred Talents Program.
Link to the paper: http://www.pnas.org/content/early/2018/03/21/1800719115
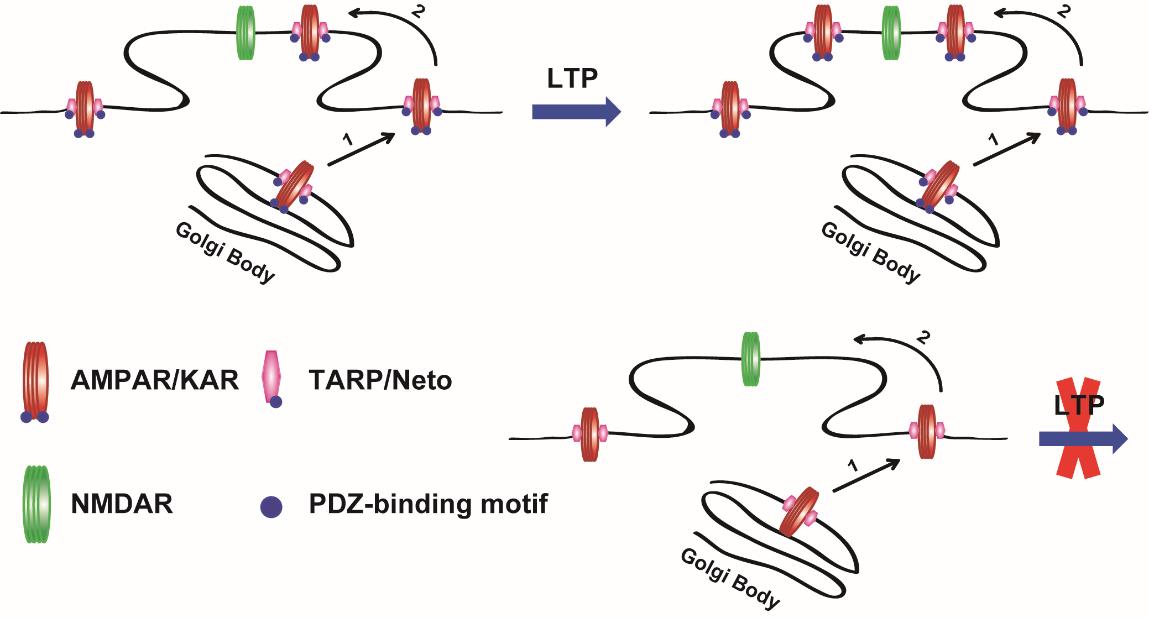
Working Model of postsynaptic PDZ-domain interactions with glutamate receptor/auxiliary protein complexes for LTP expression. (Image by Dr. SHENG Nengyin’s Group)
By SHENG Nengyin, Editor: HE Linxi
Contact:
HE Linxi
helinxi@mail.kiz.ac.cn
