Centipede rapidly subdues Kunming mouse
This study uncovers the molecular mode of action of centipede venom, which helps the tiny creatures subdue larger-bodied prey. Centipedes are known to subdue large prey by using potent, broad-acting venom. However, venom synthesis requires substantial metabolic investment, and the mechanisms of action of centipede venoms remain unclear.
LAI Ren from Kunming Institue of Zoology, Chinese Academy of Sciences, and colleagues report that in an encounter reminiscent of David and Goliath a golden head centipede (Scolopendra subspinipes mutilans) weighing around 3 g can set upon and subdue a caged laboratory mouse weighing around 45 g within 30 seconds, a single bite of the centipede injecting an estimated 30 μl of crude venom into the mouse.
Biochemical analysis of centipede venom uncovered a previously uncharacterized peptide toxin, dubbed Ssm Spooky Toxin (SsTx), that strongly inhibits KCNQ family potassium ion channels, in particular KCNQ4, which controls pulmonary vascular tone and arterial tension. Structural and functional analysis of this interaction revealed that two specific interacting residue pairs (K13-D266 and R12-D288) are crucial to the toxin's effect on KCNQ4.
In vivo tests further revealed that SsTx is the major vasoconstricting principle in venom, and that the toxin reduces respiratory rate and triggers hippocampal seizures in mice as well as inducing vessel spasms, acute hypertension, and myocardial ischemia in macaque monkeys. Most of the toxin-induced effects could be reversed by the channel-opening compound retigabine, which is approved for epilepsy treatment.
The findings reveal the molecular targets of centipede venom and point to an antidote with clinical promise.
LUO Lei, LI Bowen (Kunming Institute of Zoology, CAS), WANG Sheng (Huazhong University of Science and Technology) and WU Fangming (University of Science and Technology of China) are the co-first authors of this paper. ZHOU Ming (Baylor College of Medicine), TIAN Changlin (University of Science and Technology of China), YANG Shilong and LAI Ren (Kunming Institute of Zoology, CAS) are the corresponding authors.
This work was supported by funding from the National Science Foundation (NSF) of China, Chinese Academy of Sciences (CAS) and Yunnan Province.
Link to the article:http://www.pnas.org/content/early/2018/01/12/1714760115.full.pdf
Link to Science News: https://www.sciencenews.org/blog/science-ticker/centipede-bite-epilepsy-drug?tgt=nr
Link to Science Magazine:http://www.sciencemag.org/news/2018/01/giant-centipedes-can-kill-prey-15-times-bigger-they-are-thanks-critical-toxin
Link to Washington Post:https://www.washingtonpost.com/news/speaking-of-science/wp/2018/01/22/centipedes-eat-animals-15-times-their-size-thanks-to-this-powerful-toxin-study-finds/?utm_term=.696fb0dedf09
Link to Newsweek:http://www.newsweek.com/killer-centipede-murders-mice-thirty-seconds-spasm-inducing-spooky-toxin-fatal-786996
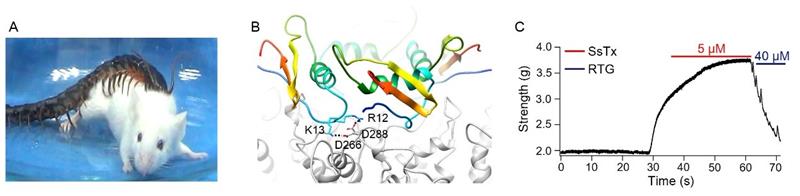
A) Image of a S. subspinipes mutilans preying on a Kunming mouse. B) Molecular docking of SsTx onto KCNQ4. The side chains of 12R/13K in SsTx and D266/D288 in KCNQ4 are shown. (C) Representative vascular contractility of thoracic aorta when challenged sequentially with 5 μM SsTx and 40 μM RTG.(Image from LAI Ren’s Group)
By YANG Shilong; Editor: HE Linxi
Contact:
HE Linxi
helinxi@mail.kiz.ac.cn

